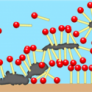
Nonionic surfactants are more EO-based and are commonly referred to as autoxyl surfactants. Nonionic surfactants are formed by forming hydrogen bonds from water-soluble hydrophilic sections. As the temperature rises, large pieces of non-ionic surfactant eventually separate at a certain point. These types of surfactants, although not ionized in water, contain both hydrophobic groups.
The temperature at which phase separation occurs is considered the limit point. Surfactant molecules are broken down in the oil phase above the limit point, and a large amount of water in the oil phase dissolves near this temperature. The boundary point increases almost linearly with n: the longer the polyoxyl ethylene chain, the greater the limit value. Nonionic surfactants used in asphalt emulsions generally have higher points above the boiling point of water, which can only occur at high temperature asphalt emulsions.
Nonionic surfactants have the advantage of being compatible with other types of surfactants. Due to their non-ionic nature, these surfactants do not show much adsorption on precipitation surfaces. In terms of unwanted loss of surfactant in the solid and liquid interface, this means that more surfactant is available to saturate the liquid-vapor interface created by mixing air bubbles, and therefore less surfactant compared to similar ionic surfactants. Are required.
Nonionic surfactants are commonly used in detergents and dishwashers. They are the second most widely used surfactant after anionics. These molecules are safe and generally more effective than anions, but for some people they cause very little skin irritation. Therefore, most cleaning products mix anionic and non-ionic surfactants to balance the cleaning potential with the risk of skin irritation.
The source of this article is the Persian section of the site, which you can read if you are fluent in Persian: سورفکتانت های نانیونیک
Exonyl CAC
Product Name: Exonyl CAC
persian Title: CAC اکسونيل
Manufacturer: Iran
Chemical Form: Liquid
Unit of measurement: kg
Package Type: 250kg barrel
Grade: CAC
Exonyl LC
Product Name: Exonyl LC
persian Title: LC اکسونيل
Manufacturer: Iran
Chemical Form: Liquid
Unit of measurement: kg
Package Type: 250kg barrel
Grade: LC
Iramid CD
Product Name: Iramid CD
persian Title: CD ايراميد
Manufacturer: Iran
Chemical Form: Liquid
Unit of measurement: kg
Package Type: Barrel
Grade: CD
Iramide CM
product Name: Iramide CM
English Title: CM ايراميد
Manufacturer: Iran
Chemical Form: Liquid
Unit of measurement: kg
Package Type: Barrel
Degree: CM
Iramide VD
Product Name: Iramide VD
English Title: VD ايراميد
Manufacturer: Iran
Chemical Form: Liquid
Unit of measurement: kg
Package Type: Barrel
Grade: VD
Irapon AL
Product Name: Irapon AL
English Title: AL ايراپون
Manufacturer: India
Chemical Form: Solid
Unit of measurement: kg
Package Type: Bag
Grade: AL
Irapon EGMS
Product Name: Irapon EGMS
persian Title: EGMS ايراپون
Manufacturer: India
Chemical Form: Solid
Unit of measurement: kg
Package Type: Bag
Grade: EGMS
Irapon GC
Product Name: Irapon GC
persian Title: GC ايراپون
Manufacturer: India
Chemical Form: Solid
Unit of measurement: kg
Package Type: Bag
Grade: GC
Irapon TS
Product Name: Irapon TS
persian Title: TS ايراپون
Manufacturer: India
Chemical Form: Solid
Unit of measurement: kg
Package Type: Bag
Grade: TS
Irapon TSS
Product Name: Irapon TSS
persian Title: TSS ايراپون
Manufacturer: India
Chemical Form: Solid
Unit of measurement: kg
Package Type: Bag
Grade: TSS